


Coninno was invited to participate in the 23rd China International Exhibition on Laboratory Medicine and Blood Transfusion Instruments and Reagents
On the morning of March 21, 2026, the 23rd China International Exhibition on Clinical Laboratory and Blood Transfusion Instruments & Reagents & the 6th China International Exhibition on IVD Upstream R21
2026-03
Live from the event! Coninno was invited to participate in the 2026 Beijing-Tianjin-Hebei Pharmaceutical, Medical Device, and IVD Elite Summit
Currently, as the centralized bulk procurement of pharmaceuticals and medical supplies is being institutionalized and normalized, and as national policies on negotiated drug and medical device pricing18
2026-03
Live from the event! Coninno was invited to participate in the 7th Medical Diagnosis Expo 2026
Over the past year, primary healthcare has continued to navigate a complex landscape. Will the implementation of AI, specialization in specific fields, or operational restructuring drive a leap in cap05
2026-03
[Allergen-Specific IgE Antibody Test Kit for Inhalant and Food Group 2 Allergens] has been officially approved!
On February 3, 2026, the “Inhalant and Food Group 2 Allergen-Specific IgE Antibody Test Kit (Fluorescent Immunoassay)” developed by Suzhou Coninno Biotechnology Co., Ltd. received a Class II Medical D03
2026-02
[Allergen-specific IgE Antibody Detection Kit for Set 1 (Inhalation and Food) Allergens (Fluorescence Immunoassay)] Officially Approved!
On October 11, 2025, the Allergen-specific IgE Antibody Detection Kit for Set 1 (Inhalation and Food) Allergens (Fluorescence Immunoassay) developed by Suzhou Coninno Biotechnology Co., Ltd. obtained the Class Ⅱ Medical Device Registration Certificate from the Jiangsu Products Administration!11
2025-10
Fluorescent Immunochromatographic Analyzer Officially Approved!
On September 2, 2025, the Fluorescent Immunochromatographic Analyzer developed by Suzhou Coninno Biotechnology Co., Ltd. obtained the Class Ⅱ Medical Device Registration Certificate from the Jiangsu Products Administration!02
2025-09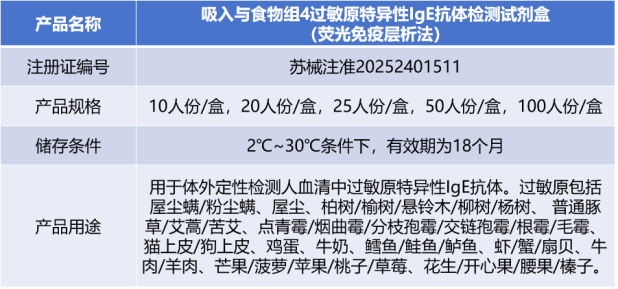
[Allergen-specific IgE Antibody Detection Kit for Set 4 (Inhalation and Food) Allergens (Fluorescence Immunoassay)] Officially Approved!
On August 1, 2025, the Allergen-specific IgE Antibody Detection Kit for Set 4 (Inhalation and Food) Allergens (Fluorescence Immunoassay) developed by Suzhou Coninno Biotechnology Co., Ltd. obtained the Class Ⅱ Medical Device Registration Certificate from the Jiangsu Products Administration!01
2025-08
[Allergen-specific IgE Antibody Detection Kit for Set 3 (Inhalation and Food) Allergens (Fluorescence Immunoassay)] Officially Approved!
On July 31, 2025, the Allergen-specific IgE Antibody Detection Kit for Set 3 (Inhalation and Food) Allergens (Fluorescence Immunoassay) developed by Suzhou Coninno Biotechnology Co., Ltd. obtained the Class Ⅱ Medical Device Registration Certificate from the Jiangsu Products Administration!31
2025-07
[Sample Diluent] Officially Approved!
On July 4, 2025, the Sample Diluent developed by Suzhou Coninno Biotechnology Co., Ltd. obtained the Medical Device Filing Certificate from the Jiangsu Provincial Medical Products Administration!04
2025-07
About Us
Product Center
News Center
Application Scenarios
Contact Us
COPYRIGHT 2025 Coninno Biotechnology All rights reserved 苏ICP备2025158326号-1 (X)网药械信息备字(XXXX)第 XXXX 号
Online Map